Causes & Triggers
The cause of psoriasis is not fully understood. Psoriasis plaques occur because the overactive immune system speeds up skin cell growth. Normal skin cells completely grow and shed (fall off) in a month. With psoriasis, skin cells do this in only three or four days. Instead of shedding, the skin cells pile up on the surface of the skin.
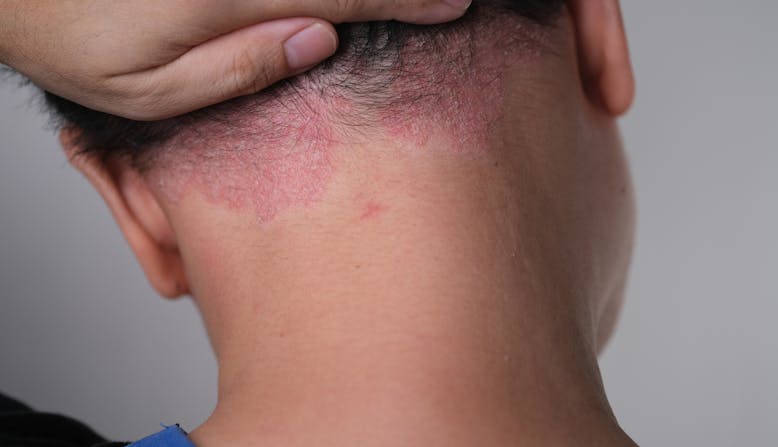
Inflammation caused by psoriasis can impact other organs and tissues in the body. People with psoriasis may also experience related health conditions. One in three people with psoriasis may also develop psoriatic arthritis. Scalp psoriasis may be an indicator of PsA, as many people have both. If you think you have scalp psoriasis, see a dermatologist to diagnose scalp psoriasis and visit a rheumatologist to screen for psoriatic arthritis.
Scalp psoriasis can be triggered by the same factors as other types of psoriasis. Common psoriasis triggers include:
- Stress
- Certain medications
- Smoking tobacco
- Alcohol
- Injuries or trauma to the skin
- Illness, such as infections
- Changes in weather
- Diet
Learn more about triggers and flares by downloading your free Flare Guide and Symptom Tracker.
Diagnosis
A health care provider will take several factors into consideration when making a diagnosis for psoriasis, including:
- The appearance of the skin. Different types of psoriasis have different appearances.
- The location of psoriasis. Certain types of psoriasis are more common in certain locations.
- Itch of the skin. This is a common symptom of psoriasis.
- A skin biopsy (the removal of a small piece of skin to be looked at under a microscope) may be needed if the diagnosis is not clear from visual inspection. The biopsy may also be done to determine if it is psoriasis or another skin condition. However, most often, psoriasis can be identified by examining the skin and does not usually require a skin biopsy.
Treatment & Management
Although there is no cure, there are more effective psoriasis treatments today than ever before. Treating psoriasis can help improve symptoms as well as lower the risk of developing psoriasis comorbidities such as psoriatic arthritis, heart disease, obesity, diabetes, and depression.
Scalp psoriasis can be difficult to treat. It may take time to find a treatment that works well because everyone responds differently to treatments. Treatments are often combined and rotated due to concerns about side effects or because treatments have stopped working after repeated use.
Treatments for psoriasis include:
%20scalp%20pso%2029.jpg?ixlib=gatsbyFP&auto=format%2Ccompress&fit=max&rect=0%2C12%2C745%2C428&w=778&h=447)
%20scalp%20Psoriasis_28.jpg?ixlib=gatsbyFP&auto=format%2Ccompress&fit=max&rect=0%2C43%2C482%2C277&w=778&h=447)


.png?ixlib=gatsbyFP&auto=compress%2Cformat&fit=max&rect=12%2C0%2C1175%2C675&w=778&h=447)