Treatment & Management
Although there is no cure, there are more effective psoriasis treatments today than ever before. Treating psoriasis can help improve symptoms as well as lower the risk of developing psoriasis comorbidities such as psoriatic arthritis, heart disease, obesity, diabetes, and depression.
Treatments for psoriasis include:
Complementary and Integrative Medicine
Complementary and integrative medicine (CIM), which includes natural products, lifestyle changes, and mind and body practices, is a popular addition to some people’s treatment plans. Talk with your health care provider before making any changes to your treatment plan to decide if it is right for you.
Prevention
There is no known way to prevent the onset of psoriasis, however, there are many ways that you can manage your triggers to reduce flares.
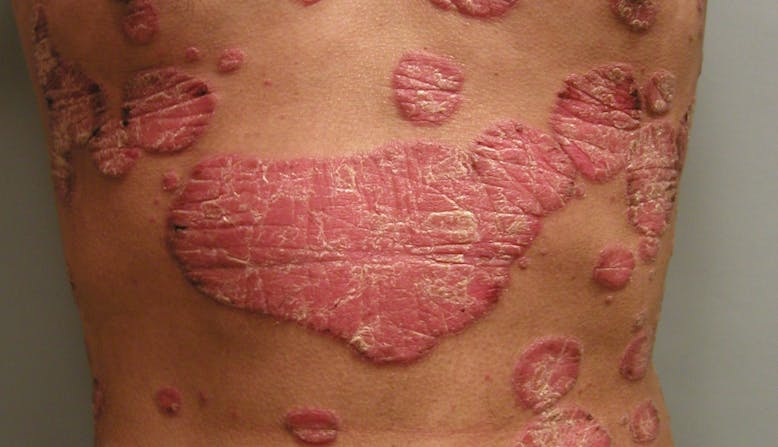
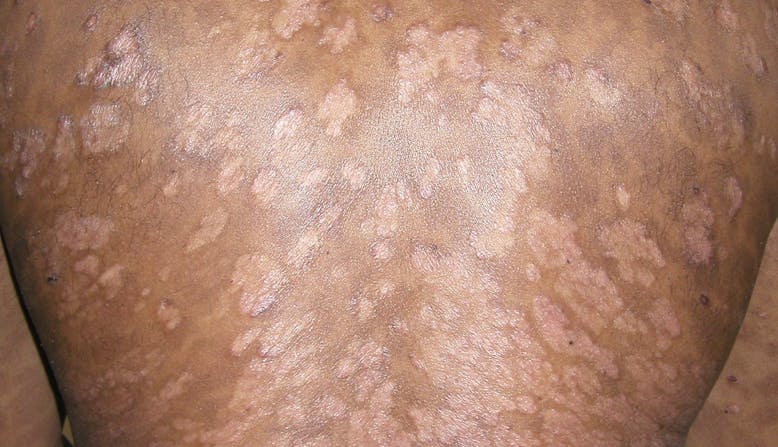
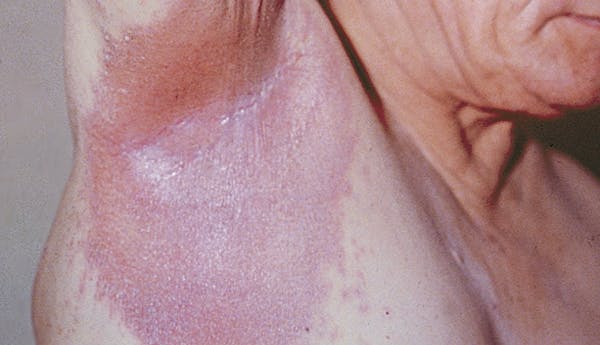
Outlook/Prognosis
Psoriatic disease is lifelong, and symptoms may resolve and recur throughout the lifetime.
While there is no cure for psoriasis, treatments today are more effective than ever before and research into new treatments, as well as a cure, is ongoing. Treating psoriasis can help improve symptoms and may decrease the associated inflammation that can lead to psoriasis comorbidities such as psoriatic arthritis, heart disease, and depression.



.png?ixlib=gatsbyFP&auto=compress%2Cformat&fit=max&rect=12%2C0%2C1175%2C675&w=778&h=447)
.png?ixlib=gatsbyFP&auto=compress%2Cformat&fit=max&rect=12%2C0%2C1175%2C675&w=778&h=447)
%20(7).png?ixlib=gatsbyFP&auto=compress%2Cformat&fit=max&rect=0%2C0%2C778%2C447&w=778&h=447)